Head-off Environmental Asthma in Louisiana (HEAL) Project
 HEAL was a collaborative research project conducted by the Tulane University Health Sciences Center and the New Orleans Department of Health. The HEAL project was funded by the National Institute of Environmental Health Sciences (NIEHS) and the National Institute for Minority Health and Health Disparities (NCMHD) of the NIH, the deLaski Family Foundation, and the Merck Childhood Asthma Network (MCAN).
HEAL was a collaborative research project conducted by the Tulane University Health Sciences Center and the New Orleans Department of Health. The HEAL project was funded by the National Institute of Environmental Health Sciences (NIEHS) and the National Institute for Minority Health and Health Disparities (NCMHD) of the NIH, the deLaski Family Foundation, and the Merck Childhood Asthma Network (MCAN).
The Foundation for the NIH was the umbrella organization for the public/private partnership between MCAN and NIH.
The purpose of the project was:
- To learn more about the effects of mold and other indoor allergens on children with asthma in post-Katrina New Orleans
- To implement evidence-based case management and mitigation of triggers in the home
The HEAL study involved 184 children, 4-12 years old, with moderate to severe asthma, and their families. A team of asthma education specialists and community health workers provided education and counseling on how to manage asthma in a transformed environment. HEAL interventionists used tailored case management based on the National Cooperative Inner-City Asthma Study and home visitation based on the Inner-City Asthma Study to reduce risk factors and triggers in the home. Efforts were made to maintain the fidelity of both these evidence-based interventions while paring them down in consideration of the limited resources and extraordinary disruption in the family and health care system post Katrina.
Nearly 75 percent of children with asthma in post-Katrina New Orleans were sensitive to mold, a figure nearly 25 percent higher than that for children in other U.S. cities.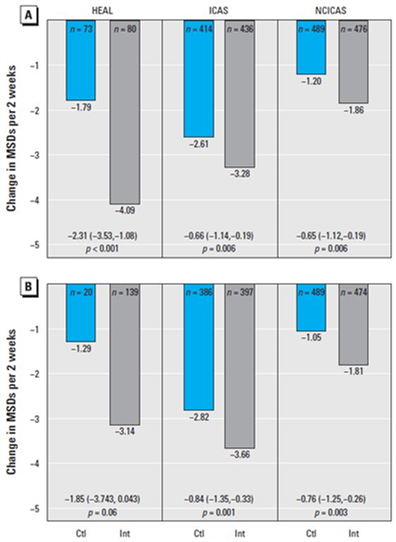 Over the 12-month intervention period, asthma counseling and mitigation of asthma triggers in the home improved asthma management, and patients reported that the average number of maximum symptom days (MSDs) declined from 6.5 to 3.6, a statistically significant 45 percent reduction. These findings were consistent with the changes observed in the NCICAS and ICAS interventions (35 percent and 62 percent reduction in symptom days, respectively).
Over the 12-month intervention period, asthma counseling and mitigation of asthma triggers in the home improved asthma management, and patients reported that the average number of maximum symptom days (MSDs) declined from 6.5 to 3.6, a statistically significant 45 percent reduction. These findings were consistent with the changes observed in the NCICAS and ICAS interventions (35 percent and 62 percent reduction in symptom days, respectively).
Learn more about the design, implementation and final outcomes of HEAL in the publications listed below:
